.jpg)
10 Best Clinical Trial Management Software (2026 List)
Healthcare organizations searching for the best clinical trial data management software in 2026 often rely on modern platforms that combine operational oversight, analytics, and regulatory compliance in one environment.
Leading solutions such as Medidata Rave, Veeva Vault, and Oracle Siebel help research sponsors coordinate global studies, while newer customizable platforms enable faster internal tool development and workflow automation. Choosing the right system depends on trial complexity, data governance requirements, and integration with existing healthcare infrastructure.
Top clinical trial platforms – quick picks
- Medidata Rave – best for global enterprise trials.
- Veeva Vault – best unified clinical operations suite.
- UI Bakery – best custom clinical operations dashboards.
- Oracle Siebel CTMS – best for compliance-heavy enterprises.
- Florence eBinders – best investigator site workflows.
- Castor EDC – best decentralized research programs.
- ClinCapture – best flexible deployment model.
- TrialKit – best patient-centric mobile trials.
How we evaluated clinical research data management software
To create this ranking of clinical research data management software, we assessed platforms based on operational automation, security controls, reporting capabilities, and integration flexibility. Particular attention was given to how tools support clinical data validation software workflows such as query management, audit trails, and monitoring dashboards. We also evaluated usability for coordinators and scalability for large multi-site research programs.
Comparison table
Medidata Rave – clinical trial data analysis software
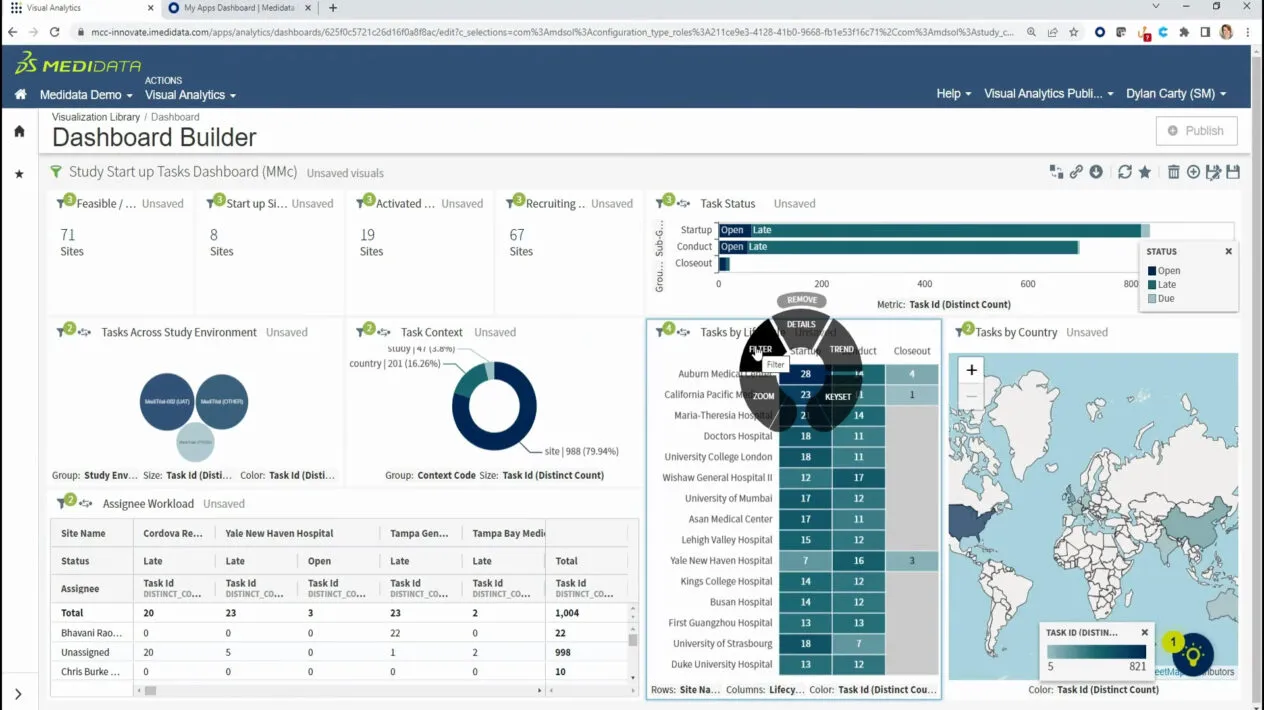
Medidata Rave is widely used as enterprise-grade clinical trial data analysis software that enables research sponsors to manage complex global trials with strong operational visibility. The platform integrates electronic data capture, study monitoring, and analytics into one ecosystem, helping teams reduce manual coordination and accelerate decision-making.
Advanced dashboards allow stakeholders to track recruitment trends, site performance, and protocol deviations in real time. This centralized approach supports faster regulatory submissions and improved study outcomes.
Key strengths:
- Advanced predictive analytics.
- Integrated monitoring workflows.
- Global scalability.
Best for: large pharmaceutical companies running multi-country trials.
Possible limitations: custom pricing and complex implementation requirements.
Veeva Vault – clinical data management system software
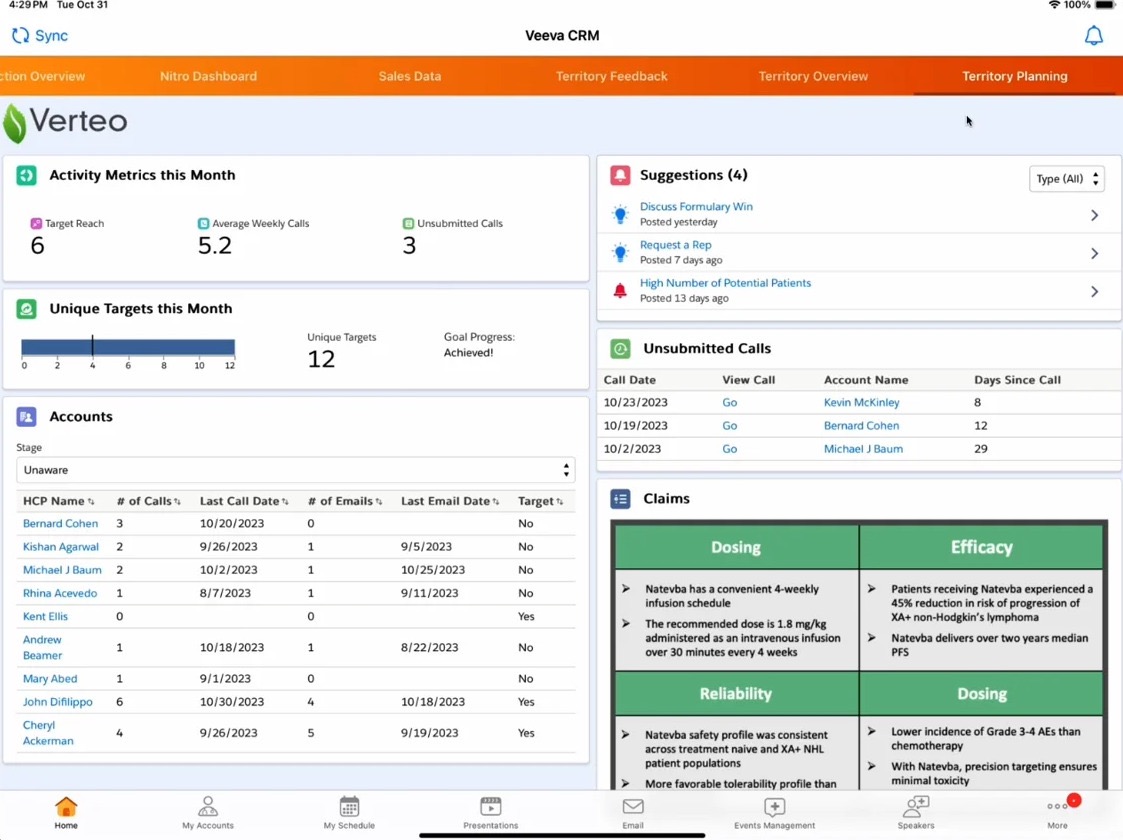
Veeva Vault provides a unified clinical data management system software environment designed to connect clinical operations, quality processes, and regulatory documentation. Its cloud-native architecture simplifies collaboration across sponsors, CROs, and investigator sites while maintaining consistent compliance standards.
Automated milestone tracking and configurable dashboards help organizations monitor study timelines effectively. By consolidating operational data into one platform, teams gain stronger oversight of research performance.
Key strengths:
- Unified clinical and regulatory suite.
- Strong reporting automation.
- Enterprise compliance features.
Best for: sponsors seeking end-to-end clinical operations management
Possible limitations: high cost and dependency on broader Veeva ecosystem
UI Bakery – clinical trial data software for custom workflows
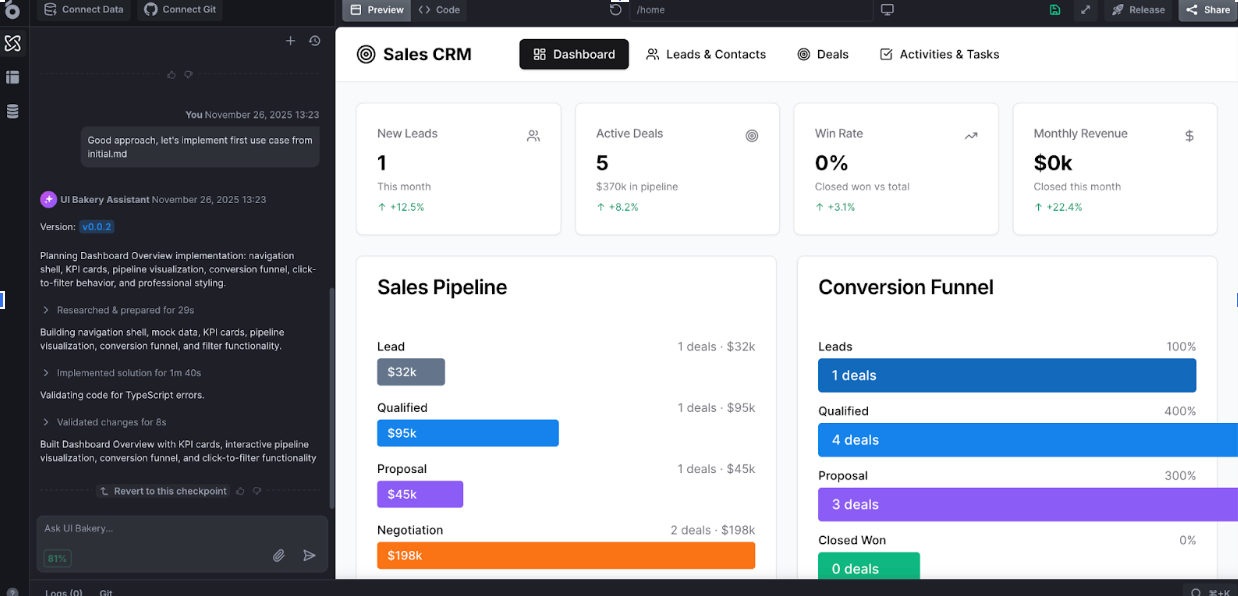
UI Bakery offers a flexible approach to clinical trial data software, enabling healthcare teams to build tailored internal dashboards, patient tracking portals, and workflow automation tools without heavy engineering investment. Instead of relying solely on rigid CTMS interfaces, research organizations can design operational apps aligned with their specific processes and integrate them directly with databases or analytics systems.
The platform supports rapid development of clinical trial data collection software interfaces for recruitment tracking, monitoring coordination, and reporting visibility. Self-hosted deployment options also help institutions meet strict security and compliance requirements.
Key strengths:
- Fully customizable research operations interfaces.
- Fast deployment of internal workflow tools.
- Integration with existing healthcare data systems.
Best for: operations teams needing flexible internal clinical coordination apps.
Possible limitations: requires process design effort and is not a traditional out-of-the-box CTMS.
Oracle Siebel – clinical data repository software
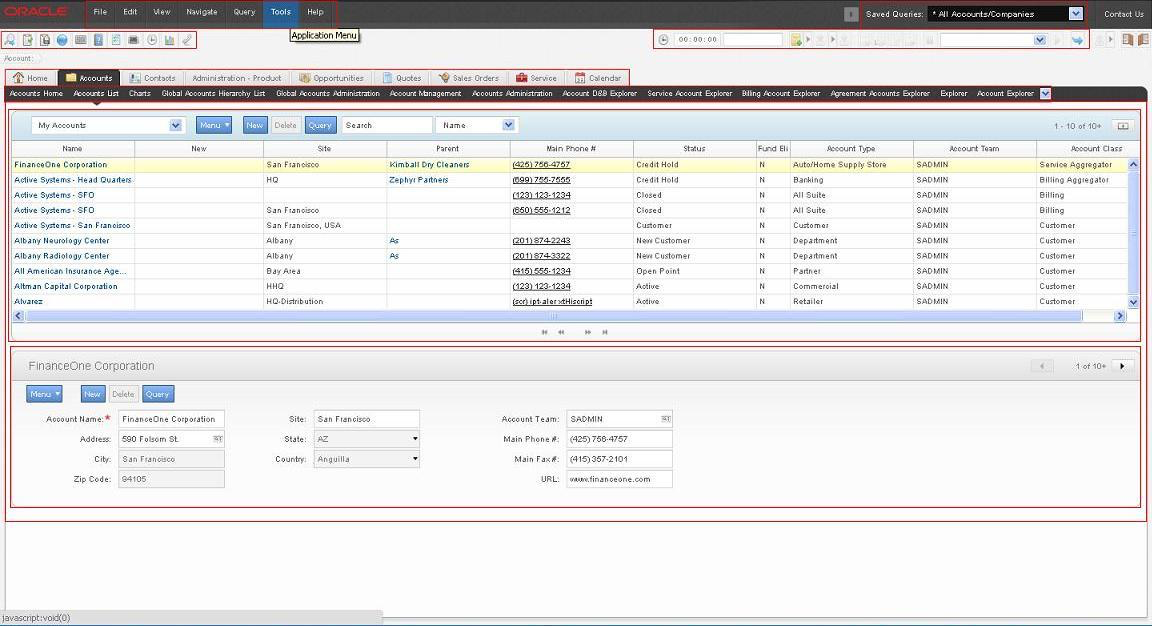
Oracle Siebel CTMS acts as a robust clinical data repository software solution designed for large regulated enterprises managing complex research portfolios. It centralizes study documentation, financial tracking, and site management data into one enterprise environment.
Deep integration with analytics tools enables structured reporting across global programs. Organizations benefit from strong governance controls that support inspection readiness and compliance monitoring.
Key strengths:
- Enterprise customization capabilities
- Strong compliance and audit features
- Integration with Oracle analytics stack
Best for: highly regulated healthcare organizations.
Possible limitations: legacy interface and longer deployment timelines.
Florence eBinders – clinical trial data entry software
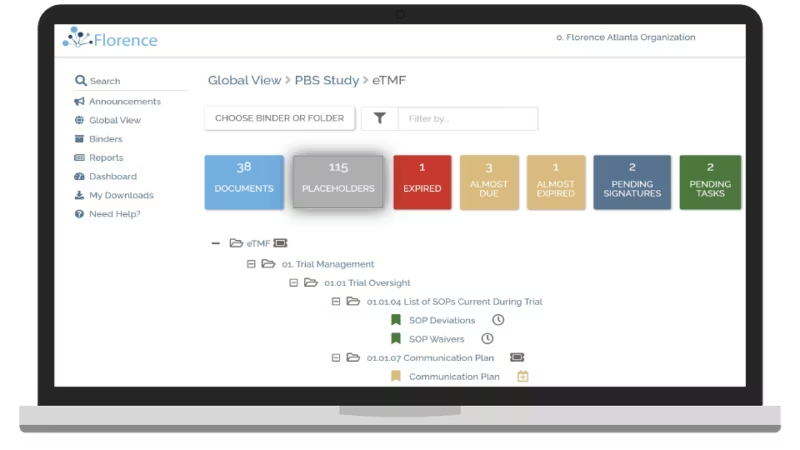
Florence eBinders digitizes regulatory binders and simplifies clinical trial data entry software workflows for investigator sites. The cloud-based system allows coordinators to manage essential documents, collaborate remotely with sponsors, and maintain real-time inspection readiness.
Electronic signatures and centralized dashboards reduce administrative burden while improving transparency. This helps research teams accelerate study startup and maintain compliance throughout execution.
Key strengths:
- Streamlined document management.
- Remote monitoring capabilities.
- Strong regulatory alignment.
Best for: clinical research sites handling multiple concurrent trials.
Possible limitations: limited operational analytics compared to full CTMS suites.
Castor – clinical trial data capture software
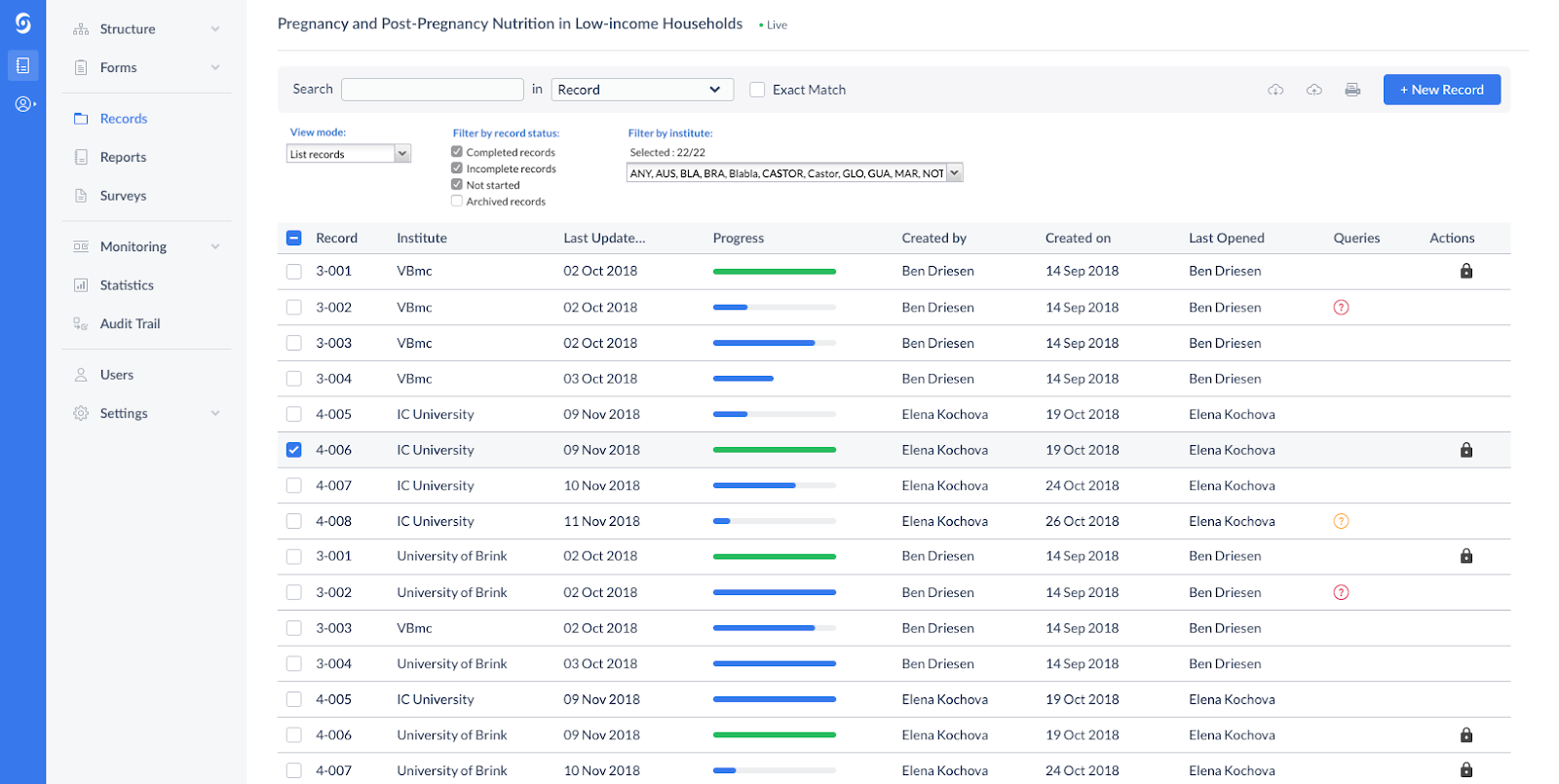
Castor provides intuitive clinical trial data capture software that supports decentralized and hybrid trial designs. Researchers can create electronic case report forms, manage enrollment workflows, and collect outcomes data through a user-friendly interface.
Built-in analytics dashboards help identify recruitment bottlenecks and protocol risks early. Its modern UX makes it easier for coordinators to manage distributed research programs.
Key strengths:
- Easy study configuration.
- Strong decentralized trial support.
- Real-time reporting.
Best for: mid-size CROs and digital-first research teams.
Possible limitations: limited enterprise customization.
ClinCapture – clinical trials data management software
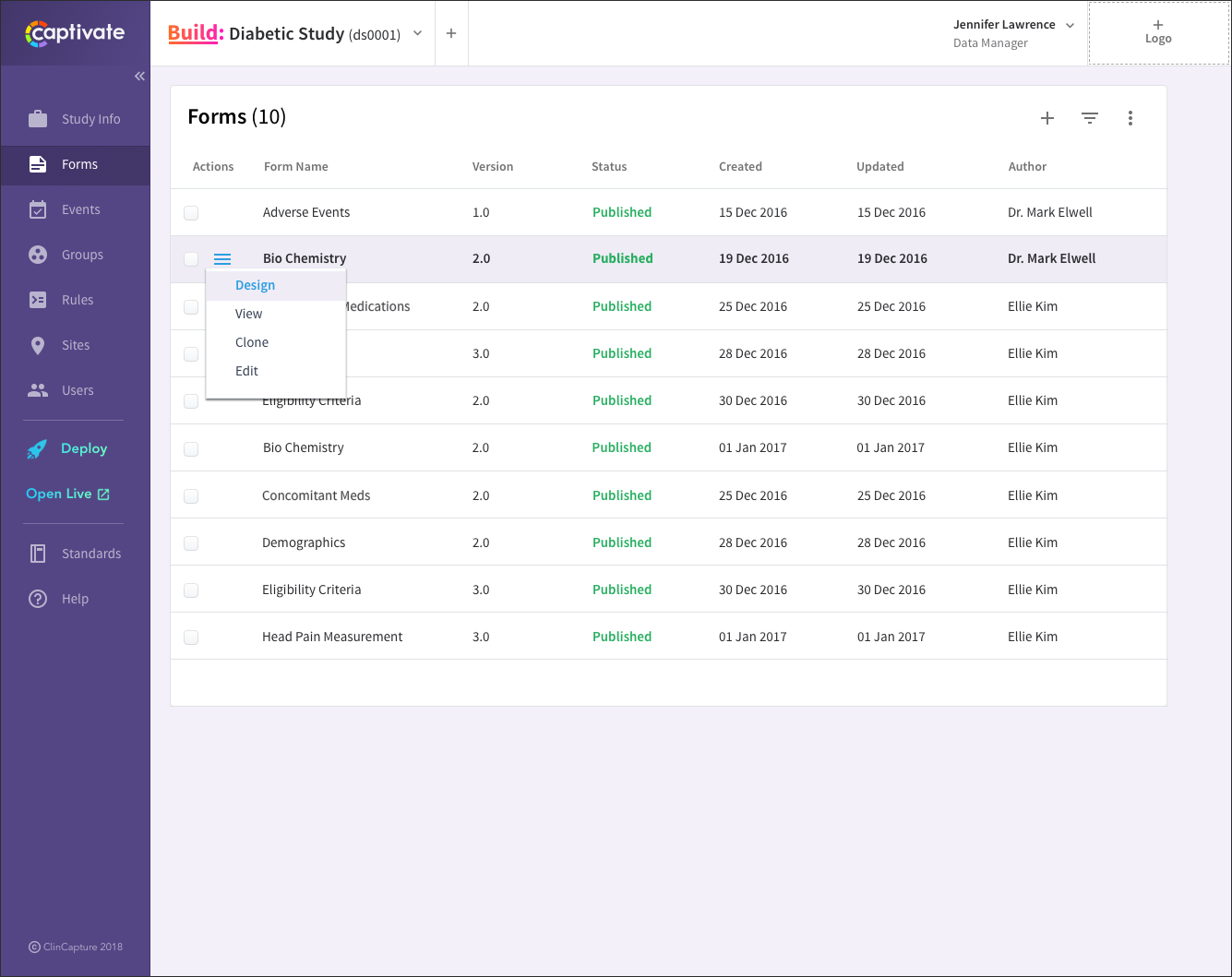
ClinCapture delivers configurable clinical trials data management software suited for organizations that need flexible hosting and customization options. The platform supports both cloud and on-premise deployments, allowing institutions to maintain control over sensitive research data.
Teams can adapt workflows and reporting structures to match specific study requirements. This flexibility makes it popular among academic research groups and smaller sponsors.
Key strengths:
- Flexible deployment models.
- Custom workflow configuration.
- Cost-effective entry point.
Best for: academic and budget-conscious research organizations.
Possible limitations: less advanced analytics compared to premium platforms.
TrialKit – clinical data analysis software
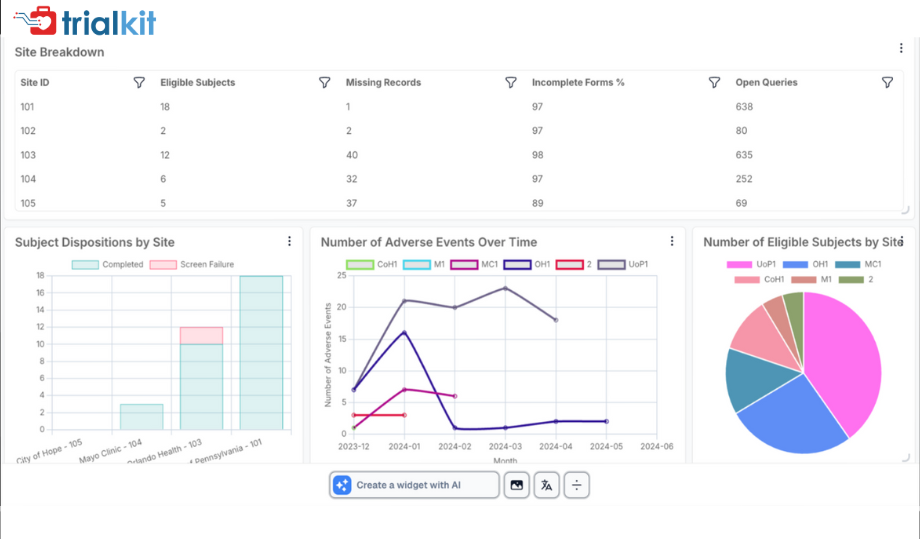
TrialKit is a mobile-first platform delivering integrated clinical data analysis software features for decentralized clinical trials. It combines electronic data capture, telehealth integration, and participant engagement tools within a single environment. Research teams gain real-time insights into study performance while improving communication with participants. Its focus on usability supports efficient execution of remote or hybrid trials.
Key strengths:
- Mobile-optimized workflows.
- Built-in telehealth capabilities.
- Participant engagement features.
Best for: sponsors running remote patient-centric trials.
Possible limitations: custom pricing transparency is limited.
How clinical data software improves research operations
Modern clinical data software solutions help research teams reduce manual coordination, improve data accuracy, and maintain compliance across study phases. Centralized dashboards and automation tools enable better collaboration between sponsors, CROs, and investigator sites.
Role of clinical data mapping software in integration
Advanced clinical data mapping software capabilities allow organizations to integrate EHR systems, laboratory platforms, and analytics tools into unified research workflows. This interoperability improves consistency and supports cross-study analysis.
Clinical data collection software and quality assurance
Using specialized clinical data collection software enables standardized reporting processes that reduce errors and improve monitoring efficiency across distributed research environments.
Final thoughts
Selecting the right clinical trial data software platform depends on study complexity, regulatory exposure, and operational scale. Enterprise sponsors often prioritize comprehensive suites, while growing research organizations benefit from flexible tools that enable faster innovation and workflow optimization.
FAQ
What is clinical data management?
Clinical data management involves collecting, validating, and maintaining research data to ensure accuracy and regulatory compliance.
How to manage clinical data?
Organizations manage clinical data through centralized systems, validation workflows, and secure repositories.
What is a clinical data repository?
A clinical data repository is a platform used to store and organize patient and study data for analysis and reporting.
How do you manage data in a clinical trial?
Teams use electronic capture systems, monitoring dashboards, and validation processes to maintain high-quality trial data.
Why is clinical data validation important?
Validation ensures research outcomes are reliable and compliant with regulatory standards.
What tools support clinical data workflows?
Modern CTMS and analytics platforms automate coordination, reporting, and operational oversight.






